Type 2 Diabetes Management Goals PowerPoint PPT Presentation
1 / 60
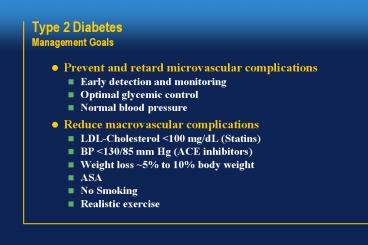
Title: Type 2 Diabetes Management Goals
1
Type 2 DiabetesManagement Goals
- Prevent and retard microvascular complications
- Early detection and monitoring
- Optimal glycemic control
- Normal blood pressure
- Reduce macrovascular complications
- LDL-Cholesterol lt100 mg/dL (Statins)
- BP lt130/85 mm Hg (ACE inhibitors)
- Weight loss 5 to 10 body weight
- ASA
- No Smoking
- Realistic exercise
2
Implementing Diabetes Treatment GoalsStrategies
- Earlier diagnosis and treatment
- Acting on results of SMBG and HbA1c
- Matching pharmacology and physiology
- Combination therapy
- Multiple oral agents (Type 2)
- Oral agents insulin or insulin analogues (Type
2) - Insulin or insulin analogues (Type 1)
3
UKPDSType 2 Diabetes . . . A Progressive
DiseaseEffect of Treatment on HbA1c
Cross-sectional, median values (7.0 vs. 7.9)
8.7
9
Conventional Intensive
8.4
8.1
8
7.5
ADA actionsuggested
7.4
Median HbA1c ()
7
ADA target
6.6
6.2 upper limit of normal range
6
0
0
3
6
9
12
15
Years From Randomization
Adapted from UK Prospective Diabetes Study
(UKPDS) Group. Lancet. 1998352837-853.
4
UKPDS THE BOTTOM LINEMain Messages for
Management of Type 2 Diabetes
- Sulfonylureas, insulin, and metformin are
effective in reducing risk of complications by
improving glycemic control - No evidence of increased risk of complications
for any single therapy - Combination therapy required almost from the
outset to achieve near-normoglycemia - Much earlier use of insulin Rx in most patients
as expected therapy in the progressive management
of Type 2 diabetes
5
How Much Combination Therapy in Type 2 Diabetes?
6
Pharmacologic Management of Type 2 Diabetes
Current and Potential Agents
- Insulin Secretagogues
- Sulfonylureas
- Glimepiride
- Glipizide
- Glyburide
- Benzoic acid
- Repaglinide
- D-Phenylalanine
- Nateglinide
- GLP-1
- Delayed Glucose Absorption
- a-Glucosidase Inhibitors
- Acarbose
- Miglitol
- Voglibose
- Insulin Sensitizers
- Metformin
- Glitazones
- Troglitazone
- Rosiglitazone
- Pioglitazone
- Others
- G-1216570
- MCC-555
- PNU-182716
- . . . and INSULIN
7
ADA Consensus on Type 2 Diabetes Therapy
Combination TherapyFrequently used or well
studied Sulfonylurea Metformin Sulfonylurea
Troglitazone Sulfonylurea Pioglitazone Sulfonylu
rea Acarbose Repaglinide Metformin Rosiglitazo
ne Metformin Pioglitazone Metformin Sulfonylur
ea Insulin Metformin Insulin Pioglitazone
Insulin Troglitazone Insulin Acarbose
Insulin Infrequently used and/orless well
studied Sulfonylurea Metformin
Troglitazone Sulfonylurea Metformin
Insulin Troglitazone Metformin Insulin
Nonpharmacologic TherapyDietExercise
Glycemic goals not achieved
Glycemic goals not achieved
Very symptomatic Severe hyperglycemia Ketosis Unre
cognized IDDM Pregnancy
Monotherapy Sulfonylureas Biguanides a-Glucosidase
inhibitors Glitazones Meglitinides Insulin
Insulin Intermediate BID Intermediate Regular
BID Multiple (3 or more) injections
Glycemic goals not achieved
Modified from Zimmerman et al. Diabetes Care.
1995.
8
Combination Therapy in Type 2 DiabetesDecision
Considerations
- HbA1c efficacy
- Reductions from baseline
- Reaching target
- Synergy of mechanisms of action
- Side effects and toxicity profile
- Frequency and severity of hypoglycemia
- Effect on weight gain
- Avoiding polypharmacy and complex regimens
- Compliance and convenience
- Cost
9
Combination Therapy in Type 2 DiabetesEstimated
Improvement
10
Type 2 DiabetesTreat to Target
- HbA1c lt7
- FBG 80-120 mg/dL (4.4 to 6.7 mmol/L)
- Postprandial BG lt180 mg/dL (10 mmol/L)
11
Practical Management of Type 2 Diabetes Mellitus
FBG gt126
All get diet and exercise
Monotherapy
126-140 mg/dL
140-200 mg/dL
200-240 mg/dL
240-300 mg/dL
gt300 mg/dL
Glitazones Metformin Acarbose
Sx
No Sx
No Sx/Sx
Sx
No Sx
Acarbose
Repaglinide
Insulin
Sulfonylurea
Sulfonylurea
Sulfonylurea
Glitazones
Metformin
Sulfonylurea
Sulfonylurea
Metformin
Oral Combination
Triple Therapy
- Evolving criteria
- If FBG gt140 mg/dL (126 mg/dL?)
- HbA1c gt8 (7?)
- Add second oral agent and titrate to maximum dose
- If no improvement
- Try a different sensitizer
- Or try triple therapy?
- Or Continue oral agent(s)and add insulin Rx at
PM or HS
12
MONOTHERAPYPlacebo-Subtracted Dose-Response to
Oral Agents Effect on HbA1c
0.5
0.5
Glimepiride
Acarbose
0.0
0.0
-0.5
-0.5
0.44
HbA1c ()
HbA1c ()
-1.0
-1.0
0.74
0.77
-1.5
-1.5
1.2
-2.0
-2.0
1.8
1.9
-2.5
-2.5
8 mg
4 mg
1 mg
300 mg
150 mg
75 mg
0.5
Metformin
0.5
Troglitazone
0.0
0.0
-0.5
-0.5
0.4
HbA1c ()
HbA1c ()
-1.0
-1.0
0.7
0.9
1.1
-1.5
-1.5
1.2
1.6
1.7
-2.0
-2.0
2.0
-2.5
-2.5
2500 mg
2000 mg
1500 mg
1000 mg
500 mg
600 mg
400 mg
200 mg
Daily Dose
Daily Dose
Data from Garber, et al. Am J Med.
1997103491-497 Goldberg, et al. Diabetes Care.
199619849-856 Precose (acarbose tablets).
Bayer Corporation. 1996 Fonseca, et al. J Clin
Endocrinol Metab. 1998833169-76.
13
MONOTHERAPYRosiglitazone Effects on HbA1c
Compared to Baseline
Compared to Placebo(Treatment Effect)
1.5
0.9
1.0
0.5
0.0
Change in Mean HbA1c ()
-0.5
-0.3
-1.0
-0.6
-1.5
-1.2
-2.0
-1.5
Placebo
4 mg
8 mg
4 mg
8 mg
Plt0.0001 Plt0.0045 Patel, et al. Diabetes.
199847(Suppl 1)A17.
14
MONOTHERAPYPioglitazone Effects on HbA1c
Previously Diet-Only Patients (Naive)
n Baseline HbA1c D from Baseline D from
Placebo Placebo 25 9.0 0.6 15
mg 26 9.9 -0.8 -1.4 30 mg 26 9.3 -0.6 -1.2
45 mg 21 9.9 -1.9 -2.5 Previously
Treated Patients n Baseline HbA1c D from
Baseline D from Placebo Placebo 54 10.9
0.8 15 mg 53 10.4 -0.1 -0.9 30
mg 59 10.4 -0.0 -0.8 45 mg 55 10.6 -0.6
-1.4
P0.05 vs. baseline Schneider et al. Diabetes.
199948 (Suppl 1)A109.
15
Monotherapy in Type 2 Diabetes MellitusSecondary
Failure
- Secondary failure rate 5 to 10 a year(UKPDS 7
a year) - Decreasing b-cell function
- Obesity
- Non-adherence to treatment
- Lack of exercise
- Intercurrent illness
16
COMBINATION THERAPYEstimated Improvements in
Glycemic Control
Regimen ? HbA1c ?
FBG Sulfonylurea metformin 1.7 65
mg/dL Sulfonylurea troglitazone 0.9-1.8 50-6
0 mg/dL Sulfonylurea pioglitazone 1.2 50
mg/dL Sulfonylurea acarbose 1.3 40
mg/dL Repaglinide metformin 1.4 40
mg/dL Pioglitazone metformin 0.7 40
mg/dL Rosiglitazone metformin 0.8 50
mg/dL Insulin oral agents Open to Target Open
to Target
DeFronzo, et al. N Engl J Med 1995333541-549
Horton, et al. Diabetes Care. 1998211462-1469
Coniff, et al. Diabetes Care. 199518817-824
Moses, et al. Diabetes Care 199922119-124
Schneider, et al. Diabetes 1999 48 (Suppl 1)
A106 Egan, et al. Diabetes 1999 48 (Suppl
1)A117. Fonseca, et al. Diabetes 199948 (Suppl
1)A100.
17
COMBINATION THERAPYSulfonylurea
MetforminEffect on HbA1c
Glyburide
Metformin
1
Metformin Glyburide
0.2
0
-0.4
- Change in Mean HbA1c ()
-1
-1.7
-2
-3
0
9
13
17
21
25
29
Treatment (wk)
Plt0.01 Plt0.001, vs. glyburide
monotherapy DeFronzo, et al. N Engl J Med.
1995333541-49.
18
COMBINATION THERAPYSulfonylurea
TroglitazoneEffect on HbA1c
2.5
Glyburide (G12)
2.0
1.5
T200
1.0
T400
0.9
T600
0.5
- Change in Mean HbA1c ()
T200 G12
0.0
T400 G12
-0.5
-0.9
T600 G12
-1.0
-1.5
-1.8
-2.0
-2.5
0
4
8
12
16
20
24
28
32
36
40
44
48
52
Treatment (wk)
Horton, et al. Diabetes Care. 1998211462-69.
19
COMBINATION THERAPYSulfonylurea
PioglitazoneEffect on HbA1c and FBG
10
1.5
0
1.0
5.6
-10
0.5
0.1
-20
0
HbA1c () D from Baseline
FBG (mg/dL) D from Baseline
-30
-0.5
-40
-1.0
-34
-0.8
-50
-1.2
-1.5
-60
-2.0
-52
PSU
PIO 15SU
PIO 30SU
PSU
PIO 15SU
PIO 30SU
P0.05 vs. baseline (paired t-test). P
Placebo PIO 15 Pioglitazone 15 mg PIO 30
Pioglitazone 30 mg SU Sulfonylurea. Schneider,
et al. Diabetes 199948 (Suppl 1)A106.
20
COMBINATION THERAPYMetformin GlitazoneEffect
on HbA1c
Metformin Pioglitazone(16 weeks)
Metformin Rosiglitazone(26 weeks)
0.6
0.6
0.4
0.2
0.3
0.3
0
0
HbA1c () D from Baseline
-0.3
-0.3
HbA1c () D from Baseline
-0.6
-0.6
-0.6
-0.6
-0.9
-0.9
-0.8
-1.2
-1.2
PMet
PIO 30Met
PMet
RSG 4 mg qd Met
RSG 8 mg qd Met
P0.05 vs. baseline (paired t-test) P0.05
vs. PMET (Dunnetts test). P Placebo MET
Metformin PIO 30 Pioglitazone 30 mg. Egan, et
al. Diabetes 199948 (Suppl 1)A117.
Plt0.0001 Fonseca, et al. Diabetes 199948
(Suppl 1)A100.
21
COMBINATION THERAPYMetformin AcarboseEffect
on HbA1c
0.2
Metformin
Metformin Acarbose
0
-0.2
P0.0001
- Mean Change in HbA1c ()
-0.4
-0.6
-0.6
-0.8
0
4
8
12
18
24
End point
Treatment (wk)
Rosenstock, et al. Diabetes Care. 1998212050-5.
22
COMBINATION THERAPYSulfonylurea and Metformin
TroglitazoneThe Bottom Line
- Baseline HbA1c 9.7
- HbA1c reduction from baseline -1.3
- Reached target HbA1c lt8 43
- Reached target HbA1c lt7 14
- Estimated cost 10/day
Yale, et al. Diabetes 199948 (Suppl 1)A118.
23
MANAGEMENT GUIDELINESCombinations of Oral
AgentsSulfonylurea-Based Regimens
- Start with Long-acting sulfonylurea (Glimepiride
or extended-release glipizide) - Add Metformin or Glitazone (If intolerance
or contraindication for metformin present)
24
MANAGEMENT GUIDELINESSecondary Failure of Oral
Combination Therapy
- Signs FPG gt140 mg/dL (gt120 mg/dL?) HbA1c gt8
(gt7?) - Causes (same as those for monotherapy) Decreas
ing ?-cell function Nonadherence to
treatment Obesity Insufficient
exercise Intercurrent illness - Rate Predictable in most patients
25
MANAGEMENT GUIDELINESCombination Therapy in Type
2 DiabetesPragmatic Approach
- Avoid polypharmacy
- Combination of insulin secretagogue insulin
sensitizer - Most simple and cost effective
- Combination of selective sensitizers
- If target FBG/HbA1c not achieved
- Try a different sensitizer or try triple therapy?
- Continue oral agent(s) Insulin Rx at PM or HS
using Insulin PenorIntensive insulin Rx alone
once daily Sulfonylurea (AM)
Metformin (PM)
26
MANAGEMENT GUIDELINESOral Agents Insulin in
Type 2 DiabetesRationale
- Simplifies insulin regimen
- Improves glycemic control
- Better patient acceptance
- Compliance
- Convenience
- Lower doses of exogenous insulin
- Less weight gain
27
COMBINATION THERAPY RATIONALEOral Agents
Insulin in Type 2 Diabetes
Synergistic or Complementary Effects
- Sulfonylureas
- Increase hepatic levels of endogenous insulin and
meet meal-related insulin requirements - Metformin
- Improves insulin sensitivity at the liver and
reduces hepatic glucose production - Glitazones
- Improve insulin action in peripheral tissues and
enhance glucose uptake - a-Glucosidase Inhibitors
- Decrease postprandial glucose absorption
28
From Insulin . . . Add-on Pills!
29
COMBINATION THERAPYInsulin TroglitazoneEffect
on HbA1c
Insulin placebo
Insulin troglitazone 200 mg
10.2
Insulin troglitazone 600 mg
9.8
9.4
-0.1
9.0
- Mean HbA1c ()
P lt0.001
8.8
-0.8
8.2
-1.4
7.8
7.4
-8
-6
-4
-2
0
1
2
4
8
10
12
16
20
24
28
Insulin
Combination Therapy
Treatment (wk)
Schwartz, et al. N Engl J Med. 1998338861-6.
30
COMBINATION THERAPYInsulin Troglitazone
Effect on HbA1c and Insulin Dose
Placebo Troglitazone 200 mg 600
mg N 118 116 116 HbA1c baseline 9.4 9.5 9.3
Change from baseline -0.1 -0.8 -1.4 Insulin
dosage () change -11 -29
Plt0.0001
Schwartz, et al. N Engl J Med 1998338961-6.
31
COMBINATION THERAPYInsulin Rosiglitazone
Effect on HbA1c and Insulin Dose
INSRSG INSRSG INSP 2 mg bid 4 mg
bid N 103 106 103 HbA1c baseline 8.9 9.1 9.0
Change from baseline 0.1 -0.6 -1.2 Insu
lin dosage () change -1 -6 -12
P0.006 vs. INSP Plt0.04 vs. INSRSG 4mg
bd. P Placebo INS Insulin RSG
Rosiglitazone.
Raskin, et al. Diabetes 199948 (Suppl 1)A94.
32
COMBINATION THERAPYInsulin Pioglitazone
Effect on HbA1c
1.0
0.5
(9.7)
(9.7)
(9.8)
-0
-0.5
HbA1c ()D from Baseline
-0.3
-1.0
-1.0
-1.5
-1.3
-2.0
-2.5
P INS
PIO 15 INS
PIO 30 INS
Plt0.05 P Placebo PIO 15 Pioglitazone 15 mg
PIO 30 Pioglitazone 30 mg INS
Insulin. Rubin, et al. Diabetes 199948 (Suppl
1)A110.
33
COMBINATION THERAPYInsulin MetforminEffect on
HbA1c
8.6
Insulin metformin
8.4
Insulin placebo
8.2
8.0
7.8
7.6
- HbA1c ()
7.4
7.2
7.0
6.8
Intensive MDI
6.6
0
2
6
Combination Therapy
Insulin
Treatment (mo)
Bergenstal, et al. Diabetes 199847(suppl 1)A89.
34
From Pills . . . Add-on Insulin!
35
COMBINATION THERAPYGlimepiride InsulinEffect
on FPG and Insulin Dosage
Placebo insulin
Glimepiride insulin titrated to FPG lt140 mg/dL
Insulin Dosage
300
FPG
100
250
75
- Mean mg/dL
200
Mean Units/Day
50
150
25
100
0
0
4
8
12
16
20
24
0
4
8
12
16
20
24
Treatment (wk)
Treatment (wk)
Plt0.001
Plt0.001
Plt0.05
Riddle, et al. Diabetes Care. 1998211052-7.
36
COMBINATION THERAPYGlimepiride InsulinEffect
on HbA1c and Insulin Dose
- Placebo Insulin Glimepiride Insulin
- Initial mean HbA1c 9.9 9.7
- Final mean HbA1c 7.7 7.6
- Final insulin dose 78 U/day 49 U/day (-38)
- Subjects needinginsulin dosegt 100 U/day 9/62
(14) 4/70 (6) - Dropouts 11/73 (15) 2/72 (3)
Plt0.001 Plt0.01, between-group difference
Data from Riddle, et al. Diabetes Care.
1998211052-7.
37
COMBINATION THERAPYSulfonylurea Lispro AC or
NPH at HS or MetforminEffects on HbA1c and Weight
Baseline HbA1c Followup HbA1c Followup Weight
12
12
10.4
10.2
10.0
10
10
-1.9
-1.9
-2.3
8
8
Weight Gain (kg)
HbA1c ()
6
6
4
4
2
2
3.4 kg
0.9 kg
2.3 kg
0
0
Su Metformin (n40)
Su NPH (n50)
Su LP (n42)
Browdoe, et al. Diabetes 199948 (Suppl 1)A104.
38
COMBINATION THERAPYThe FINFAT Study
NPH Insulin BID (n24) NPH Insulin HS Glyburide
(n22) NPH Insulin HS Metformin (n19) NPH
Insulin HS Metformin Glyburide (n23)
6
10
4
9
D Body Weight (kg)
HbA1c ()
2
8
0
7
-2
40
0
30
-1
D HbA1c ()
HS Insulin Dose (U)
20
-2
10
-3
0
3
6
9
12
0
3
6
9
12
Time (mo)
Time (mo)
Yki-Jarvinen, et al. Ann Intern Med
1999130380-96.
39
COMBINATION THERAPY The FINFAT StudyNPH Insulin
at HS Various Agents
D A1c ()
Dropouts ()
(5/24) 21
0
25
20
-1
(2/24) 8
15
(1/24) 4
NPH at HS
10
(0/24) 0
-2
1.9
2.0
Glyburide Metformin Gly Met NPH at AM
5
2.1
2.5
0
-3
D Weight (Kg)
Insulin Dose
4.6
53
5
60
3.9
3.6
4
36
40
3
24
20
2
0.9
20
1
0
0
Yki-Jarvinen, et al. Ann Intern Med
1999130380-96.
40
COMBINATION THERAPYOral Agents Inhaled Insulin
ACEffect on HbA1c
10
9
8
HbA1c ()
7
6
5
Weeks
Plt0.0001 Weiss SR, Barger S, Cheng S, et al.
Diabetes 199948 (Suppl 1)A12.
41
MANAGEMENT GUIDELINESAdding Insulin to an Oral
Agent
- Continue oral agent(s) at same dosage (eventually
reduce) - Add single, evening insulin dose (10 U) using
Insulin Pen - NPH insulin (bedtime)
- 70/30 insulin (evening meal)
- Glargine insulin (basal insulin, awaiting
approval) - Adjust dose by SMBG (fasting)
- Increase insulin dose weekly if needed
- Increase 4 U if FBG gt180 mg/dL
- Increase 2 U if FBG gt140 mg/dL
Medical Management of Type 2 Diabetes. 4th ed.
Alexandria, Va American Diabetes Association
19981-139.
42
Which Insulin is Better and Easier for
Combination Therapy?
43
Comparison of Human Insulins
Onset of Duration of Insulin Preparations
Action Peak Action Lispro 5 to 15 min 1 to 2
hr 4 to 6 hr Human Regular 30 to 60 min 2 to 4
hr 6 to 10 hr Human NPH 1 to 2 hr 4 to 6 hr 10 to
16 hr Human Lente 1 to 2 hr 4 to 6 hr 10 to 16
hr Human Ultralente 2 to 4 hr Unpredictable lt24 hr
The time course of action of any insulin may
vary in different individuals, or at different
times in the same individual. Because of
thisvariation, time periods indicated here
should be considered as general guidelines only.
44
INSULIN TACTICS Twice Daily SplitMixed Regimens
Regular
Lispro
Lispro
Lispro
Insulin Effect
Insulin Effect
Reg
Reg
NPH
NPH
NPH
NPH
B
L
S
HS
B
B
L
S
HS
B
Meals
Meals
45
INSULIN TACTICSMDI NPH Regular AC
NPH at AM and HS Regular AC
NPH at HS Regular AC
Reg
Reg
Insulin Effect
Reg
Reg
Reg
Insulin Effect
NPH
NPH
NPH
B
L
S
HS
B
B
L
S
HS
B
Meals
Meals
46
INSULIN TACTICSMDI NPH Lispro AC
NPH at AM and HS Lispro AC
NPH at HS Lispro AC
Lispro
Lispro
Lispro
Lispro
Lispro
Insulin Effect
Insulin Effect
NPH
NPH
NPH
B
L
S
HS
B
B
L
S
HS
B
Meals
Meals
47
INSULIN TACTICSMDI Ultralente at PM Regular AC
48
INSULIN TACTICS Human Ultralente Insulin A True
Basal Insulin?
- N 5 subjects with type 1 diabetes
- Onset of action 2 to 4 hours
- Broad variable peak between 6 and 12 hrs after
injection
This pattern, and the duration of action of
Human Ultralente, appear to more closely mimic
those after injection of animal NPH or Lente than
Beef Ultralente.
Freeman SL, OBrian PC, Rizza RA Diab Res Clin
Pract 12187, 1991
49
INSULIN TACTICSMDI The Quest for a Basal Insulin
Combinations of Lispro/NPH AC and NPH at HS
Lalli et al. Diabetes Care. 199922468-477.
50
INSULIN TACTICS The Ideal Basal Insulin
- Mimics normal pancreatic basal insulin secretion
- Long-lasting effect
- Smooth peakless profile
- Reproducible and predictable effects
- Reduced nocturnal hypoglycemia
- Once-daily administration for convenience
- Pharmacodynamic effects similar to insulin pump
51
INSULIN TACTICSInsulin GlargineA New
Long-acting Insulin Analogue
- Modifications to human insulin chain
- Substitution of glycine at position A21
- Addition of two arginines at position B30
- Gradual release pattern from injection site
52
INSULIN TACTICSGlargine Vs. NPH Insulin in Type
1 DMAction Profiles by Glucose Clamp
6
Glargine insulin NPH insulin
5
4
3
Glucose Utilization Rate (mg/kg/h)
2
1
0
0
30
20
10
End of observation period
Time (h) after S.C. Injection
Lepore, et al. Diabetes 199948 (Suppl 1)A97.
53
INSULIN TACTICS Glargine vs. NPH Insulin in
Nondiabetic SubjectsPharmacokinetics by Glucose
Clamp
Linkeschowa R, et al. Diabetes.199948(Suppl
1)A97.
54
INSULIN TACTICS The Basal/Bolus Insulin Concept
- Basal Insulin
- Insulin requirement to suppress hepatic glucose
production between meals - Bolus Insulin (prandial)
- Insulin requirement to maintain normal glucose
disposal after eating - Ideally, each component should come from
a different insulin with a specific
profile - CSII best model of the Basal/Bolus concept
55
INSULIN TACTICS Insulin Glargine in Type 2
Diabetes
- Baseline HbA1c 8.5
- Similar HbA1c improvement (? 0.4 to 0.6) with
less nocturnal hypoglycemia and less weight gain
in the insulin Glargine group
Wt. Gain Nocturnal Hypos NPH 3.1
lb 40 Glargine 0.9 lb 31
Plt0.01 P lt0.02
Rosenstock, et al. Diabetes 48(Suppl 1)A100, 1999
56
The Potential Role of Basal Insulin
57
INSULIN TACTICS THE FUTUREMDI Glargine at HS
Inhaled Insulin AC
Inhaled Inhaled Inhaled Insulin
Insulin Insulin
Glargine
Insulin Effect
(HOE-901)
B
L
S
HS
B
Meals
58
Combination Oral Agents InsulinStarting
Insulin . . .
59
Evening NPH or Ultralente or 70/30 or Glargine?
60
Bridge Therapy