Gated Transport through Carbon Nanotube Membranes NIRT CBET-0709090
1 / 1
Title:
Gated Transport through Carbon Nanotube Membranes NIRT CBET-0709090
Description:
Sangil Kim1,2, Francesco Fornasiero1, Michael Stadermann1, Alexander Chernov1, ... Colvin4, Aleksandr Noy1,4, Olgica Bakajin,1,2 and Costas P. Grigoropoulos3 ... –
Number of Views:81
Avg rating:3.0/5.0
Title: Gated Transport through Carbon Nanotube Membranes NIRT CBET-0709090
1
Gated Transport through Carbon Nanotube
MembranesNIRT CBET-0709090
Sangil Kim1,2, Francesco Fornasiero1, Michael
Stadermann1, Alexander Chernov1, Hyung Gyu Park1,
Jung Bin In3, Ji Zang5, David Sholl5, Michael
Colvin4, Aleksandr Noy1,4, Olgica Bakajin,1,2 and
Costas P. Grigoropoulos3 1 Physical and Life
Sciences, LLNL 2 NSF Center for Biophotonics,
UC Davis 3Mechanical Engineering, UC Berkeley
4School of Natural Sciences, UC Merced,
5Chemical and Biochemical Engineering, Georgia
Tech
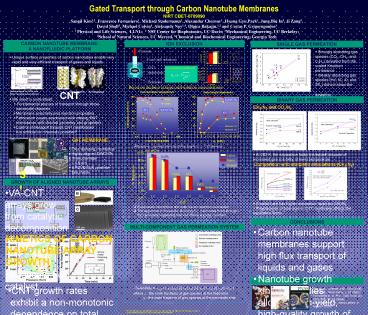
CARBON NANOTUBE MEMBRANEA NANOFLUIDIC PLATFORM
- Strongly absorbing gas species (CO2, CH4, and
C2H4) deviated from the scaled Knudsen permeance - Weakly absorbing gas species (He, N2, Ar, and
SF6) did not show the deviation.
- Unique surface properties of carbon nanotubes
enable very rapid and very efficient transport of
gases and liquids - We need to understand
- Fundamental physics of transport through these
nanoscale channels - Membrane selectivity and rejection properties
- Fabrication issues associated with making CNT
membranes with desired geometry and properties - Control of transport through CNT membranesAre
artificial ion channels possible?
Rejection declines at larger salt solution
concentrations
CNT
Aquaporin
Gas transport in CNTs and other nanoporous
materials
K channel
K3Fe(CN)6
CH4/N2 and CO2/N2
KCl
K3Fe(CN)6
KCl
CNT MEMBRANE
- Free standing membrane
- Highly aligned DWCNTs
- Inner diameter 1.6 nm
- LPCVD Si3N4 pinhole-free matrix
- Rejection constant when the Debye length is gtgt
CNT diameter
- At 263 K, the separation factor increased
because of increased gas solubility at lower
temperature.
Comparison with atomistic simulations (CH4/N2)
- VA-CNT arrays grow from catalytic decomposition
of carbon precursor, C2H4, over nanoscale Fe
catalyst
- Smaller tube has higher separation factor for
CH4/N2. - Polydisperse of tube size in CNT membrane
affects the separation factor.
- Electrostatic interactions dominate the ion
rejection mechanism - The largest ion in this series, Ru(bipy)3Cl2,
permeates freely through the membrane suggesting
that size effects are less important
- Carbon nanotube membranes support high flux
transport of liquids and gases - Nanotube growth kinetics studies allowed
high-yield, high-quality growth of aligned
nanotube arrays - CNT membranes show good ion rejection
characteristics - Ion rejection mechanism is based on electrostatic
repulsion and follows Donnan model predictions - Strongly absorbing gas species deviated from
Knudsen permeance due to preferential
interactions with CNTs side walls. - At low temperature gas separation factor
increased because of increased gas solubility
overall gas separation factors are still lower
than necessary for practical gas separation
KINETICS OF CARBON NANOTUBE ARRAY GROWTH
- CNT growth rates exhibit a non-monotonic
dependence on total pressure and humidity.
Optimal process pressure and water concentration
produce growth rate of 30?m/min. - Nanotube growth rate remains essentially constant
until growth reaches an abrupt and irreversible
termination. - We developed a model that predicts termination
kinetics
- Selectivity ?A/B yA/(yB) / xA/(xB)
yA/(1-yA) / xA/(1-xA) - where x the mole fractions of gas species at
the feed side - y the mole fractions of gas species
at the permeate side
- Holt et. al., Science, 312, 1034 (2006)
- Noy et. al., Nano Today, 2, 22 (2007)
- Fornasiero et. al. Proc. Natl. Acad. Sci USA,
105, 17217 (2008) - Stadermann et. al., Nano Letters, in revision
(2008)
Part of the work at LLNL was performed under the
auspices of the U.S. Department of Energy by
Lawrence Livermore National Laboratory under
Contract DE-AC52-07NA27344.