Chemistry - Matter Unit PowerPoint PPT Presentation
1 / 19
Title: Chemistry - Matter Unit
1
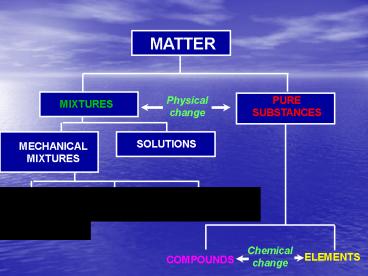
SUSPENSIONS
ORDINARY MECHANICAL MIXTURES
COLLOIDS
ELEMENTS
COMPOUNDS
2
What is a pure substance?
- A pure substance has a definite composition
(proportion). - The composition of a substance will have the same
percent of elements no matter where the sample
was obtained. - Water from the Atlantic Ocean and water from the
Pacific Ocean (once cleaned up) will have the
same composition of hydrogen to oxygen.
3
What is a pure substance?
- A pure substance, by definition, is an element or
a compound. - Salt, Coal, Gold
4
Pure Substance
Pure Substance
Compound
Element
5
What is an element?
- A substance that cannot be decomposed any farther
by simple chemical means - An element has a definite composition. Gold from
South Africa is the same, when purified, as a
sample from California.
6
Chemistry Vocabulary
- A Symbol is a representation of an element.
- One or two letters may be used.
- The first letter is upper case while the second
is lower case.
H C O Fe Cl Ag Cu Ba Pb
7
Elemental Samples
- Zinc, copper, lead, carbon, sulfur
8
What is a compound?
- A compound is a substance that is made from the
atoms of two or more elements that are chemically
bonded.
9
What is a compound?
- A compound is a substance that cannot be
decomposed any farther by simple physical means. - A compound is made up of two or more elements
chemically combined.
10
What is a compound?
- A compound no longer has the properties of its
constituent elements. - Table salt, NaCl or sodium chloride, is a
compound of the element sodium and the element
chlorine.
11
Sodium
12
Sodium metal
- Soft, can be cut with a knife
- Shiny
- Good conductor of electricity
- Very reactive
13
Sodium in water
- Observe the reaction
- What is the color of the solution
- Do you observe any gas produced
- Is it a chemical or physical reaction?
- Does the reaction terminate immediately
14
Chlorine gas
15
Chlorine gas
- Greenish gas
- Poisonous
- Heavier than air
16
Sodium in chlorine gas
17
Properties of Sodium chloride ( NaCl)
- Sodium chloride dissolves in water rather than
reacts with water. - Sodium chloride is a white solid, not a poisonous
green gas. - Sodium chloride is its own substance with its own
properties, not those of either sodium or
chlorine.
18
Samples of Other Compounds
- Sucrose (table sugar), Sodium Chloride, Water,
Copper(II) sulfate
19
Colored Compounds
- Cobalt(II) chloride, Iron(II) sulfate, Potassium
dichromate, Potassium chromate, Nickel(II)
nitrate, copper(II) sulfate