Valence Bond Theory PowerPoint PPT Presentation
Title: Valence Bond Theory
1
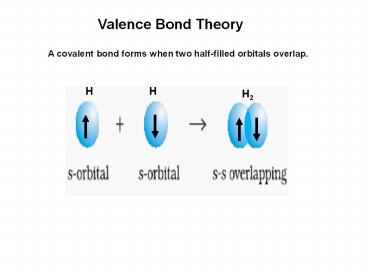
Valence Bond Theory
A covalent bond forms when two half-filled
orbitals overlap.
H
H
H2
2
Electron Configuration of C
3
Electron Promotion
90o
4
Methane
109o
5
Hybridization
A theoretical process involving the mixing of
orbitals to create a new set of orbitals. It
only occurs when an atom forms a covalent bond.
These hybrid orbitals are 109o apart.
6
Bonding in Methane
7
Ethene
120o
8
sp2 hybridization
120o
9
Bonding in Ethene
sp2 hybrid orbitals
sigma (s) bonds ? end-to-end overlap of orbitals
unhybridized p orbitals
pi (?) bond ? side-by-side overlap of orbitals
10
Acetylene
180o
11
sp hybridization
180o
12
Bonding in Acetylene
unhybridized p orbitals
sp hybrid orbitals
pi bond
sigma bond
sigma bond
pi bond
PowerShow.com is a leading presentation sharing website. It has millions of presentations already uploaded and available with 1,000s more being uploaded by its users every day. Whatever your area of interest, here you’ll be able to find and view presentations you’ll love and possibly download. And, best of all, it is completely free and easy to use.
You might even have a presentation you’d like to share with others. If so, just upload it to PowerShow.com. We’ll convert it to an HTML5 slideshow that includes all the media types you’ve already added: audio, video, music, pictures, animations and transition effects. Then you can share it with your target audience as well as PowerShow.com’s millions of monthly visitors. And, again, it’s all free.
About the Developers
PowerShow.com is brought to you by CrystalGraphics, the award-winning developer and market-leading publisher of rich-media enhancement products for presentations. Our product offerings include millions of PowerPoint templates, diagrams, animated 3D characters and more.