Introducing%20the%20P-V%20diagram - PowerPoint PPT Presentation
Title:
Introducing%20the%20P-V%20diagram
Description:
Introducing the P-V diagram P-V (pressure versus volume) diagrams can be very useful. What are the units resulting from multiplying pressure in kPa by volume in liters? – PowerPoint PPT presentation
Number of Views:215
Avg rating:3.0/5.0
Title: Introducing%20the%20P-V%20diagram
1
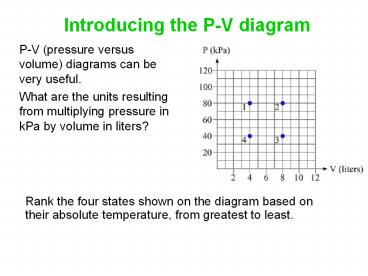
Introducing the P-V diagram
- P-V (pressure versus volume) diagrams can be very
useful. - What are the units resulting from multiplying
pressure in kPa by volume in liters?
Rank the four states shown on the diagram based
on their absolute temperature, from greatest to
least.
2
Introducing the P-V diagram
- P-V (pressure versus volume) diagrams can be very
useful. - What are the units resulting from multiplying
pressure in kPa by volume in liters?
Rank the four states shown on the diagram based
on their absolute temperature, from greatest to
least. Temperature is proportional to PV, so rank
by PV 2 gt 13 gt 4.
3
Isotherms
- Isotherms are lines of constant temperature.
- On a P-V diagram, isotherms satisfy the equation
- PV constant
4
Thermodynamics
- Thermodynamics is the study of systems involving
energy in the form of heat and work. - Consider a cylinder of ideal gas, at room
temperature. - When the cylinder is placed in a
- container of hot water, heat is
- transferred into the cylinder.
- Where does that energy go?
- The piston is free to move up
- or down without friction.
5
Thermodynamics
6
The First Law of Thermodynamics
- Some of the added energy goes into raising the
temperature of the gas (we call this raising the
internal energy). The rest of it does work,
raising the piston. Conserving energy - (the first law of thermodynamics)
- Q is heat added to a system (or removed if it is
negative) - is the internal energy of the system (the
energy associated with the motion of the atoms
and/or molecules), so is the change in
the internal energy, which is proportional to the
change in temperature. - W is the work done by the system.
- The First Law is often written as
7
Work
- We defined work previously as
- (true if the force is constant)
- F PA, so
- At constant pressure the work done by the system
is the pressure multiplied by the change in
volume. - If there is no change in volume, no work is done.
- In general, the work done by the system is the
area under the P-V graph. This is why P-V
diagrams are so useful.
8
Work the area under the curve
- The net work done by the gas is positive in this
case, because the change in volume is positive,
and equal to the area under the curve.
9
A P-V diagram question
An ideal gas initially in state 1 progresses to a
final state by one of three different processes
(a, b, or c). Each of the possible final states
has the same temperature. For which process is
the change in internal energy larger? 1. a
2. b 3. c 4. Equal for all three 5. We cant
determine it
10
A P-V diagram question
- Because the change in temperature is the same,
the change in internal energy is the same for all
three processes.
11
Another P-V diagram question
An ideal gas initially in state 1 progresses to a
final state by one of three different processes
(a, b, or c). Each of the possible final states
has the same temperature. For which process is
more heat transferred into the ideal gas? 1.
a 2. b 3. c 4. Equal for all three 5. We
cant determine it
12
Another P-V diagram question
- The heat is the sum of the change in internal
energy (which is the same for all three) and the
work (the area under the curve), so whichever
process involves more work requires more heat.
13
Another P-V diagram question
- The heat is the sum of the change in internal
energy (which is the same for all three) and the
work (the area under the curve), so whichever
process involves more work requires more heat. - Process c involves more
- work, and thus requires
- more heat.
14
Constant volume vs. constant pressure
We have two identical cylinders of ideal gas.
Piston 1 is free to move. Piston 2 is fixed so
cylinder 2 has a constant volume. We put both
systems into a reservoir of hot water and let
them come to equilibrium. Which statement is
true? 1. Both the heat Q and the change in
internal energy will be the same for the two
cylinders 2. The heat is the same for the two
cylinders but cylinder 1 has a larger change in
internal energy. 3. The heat is the same for the
two cylinders but cylinder 2 has a larger change
in internal energy. 4. The changes in internal
energy are the same for the two cylinders but
cylinder 1 has more heat. 5. The changes in
internal energy are the same for the two
cylinders but cylinder 2 has more heat.
15
Constant volume vs. constant pressure
- Each cylinder comes to the same temperature as
the reservoir. How do the changes in internal
energy compare? - Which cylinder does more work?
16
Constant volume vs. constant pressure
- Each cylinder comes to the same temperature as
the reservoir. How do the changes in internal
energy compare? - The same number of moles of the same gas
experience the same temperature change, so the
change in internal energy is the same. - Which cylinder does more work?
- Cylinder 2 does no work, so cylinder 1 does more
work. - By the first law, cylinder 1 requires more heat
to produce the same change in temperature as
cylinder 2. The heat required depends on the
process.
17
Solving thermodynamics problems
- A typical thermodynamics problem involves some
process that moves an ideal gas system from one
state to another. - Draw a P-V diagram to get some idea what the
work is. - Apply the First Law of Thermodynamics (this is a
statement of conservation of energy). - Apply the Ideal Gas Law.
- the internal energy is determined by the
temperature - the change in internal energy is determined by
the change in temperature - the work done depends on how the system moves
from one state to another (the change in internal
energy does not)
18
Constant volume (isochoric) process
- No work is done by the gas W 0. The P-V
diagram is a vertical line, going up if heat is
added, and going down if heat is removed. - Applying the first law
- For a monatomic ideal gas
19
Constant pressure (isobaric) process
- In this case the region on the P-V diagram is
rectangular, so its area is easy to find. - For a monatomic ideal gas
20
Heat capacity
- For solids and liquids
- For gases , where C, the heat
capacity, depends on the process. - For a monatomic ideal gas
- Constant volume
- Constant pressure
- In general
21
Constant temperature (isothermal) process
- No change in internal energy
- The P-V diagram follows the isotherm.
- Applying the first law, and
- using a little calculus
22
Zero heat (adiabatic) process
- Q 0. The P-V diagram is an interesting line,
given by - For a monatomic ideal gas
- Applying the first law
23
Whiteboard




![[DOWNLOAD]PDF Introducing the History of the English Language PowerPoint PPT Presentation](https://s3.amazonaws.com/images.powershow.com/10054016.th0.jpg?_=20240612129)
![[DOWNLOAD]⚡️PDF✔️ Introducing the History of the English Language PowerPoint PPT Presentation](https://s3.amazonaws.com/images.powershow.com/10052365.th0.jpg?_=20240610114)
![[PDF] READ Free ThetaHealing®: Introducing an Extraordinary PowerPoint PPT Presentation](https://s3.amazonaws.com/images.powershow.com/10084521.th0.jpg?_=20240724056)


![get⚡[PDF]❤ Introducing Second Language Acquisition (Cambridge Introductions to Language PowerPoint PPT Presentation](https://s3.amazonaws.com/images.powershow.com/10046722.th0.jpg?_=202406030512)







![[PDF] Introducing the LSAT: The Fox Test Prep Quick & Dirty LSAT Primer Paperback – December 25, 2012 Full PowerPoint PPT Presentation](https://s3.amazonaws.com/images.powershow.com/10078410.th0.jpg?_=202407130910)

![[PDF] PEDIATRIC DENTISTRY: BUILDING A NO-FEAR PRACTICE: Introducing Children to a Lifetime of Positive Dental Care Ipad PowerPoint PPT Presentation](https://s3.amazonaws.com/images.powershow.com/10085939.th0.jpg?_=20240729059)










