Photosynthesis - PowerPoint PPT Presentation
1 / 57
Title:
Photosynthesis
Description:
Two main parts (reactions) 1. Light Reaction or. Light ... Circadian rhythm ... 1. Circadian rhythm. 2. Infradian/ultradian Rhythm. 3. Circannual Rhythms ... – PowerPoint PPT presentation
Number of Views:213
Avg rating:3.0/5.0
Title: Photosynthesis
1
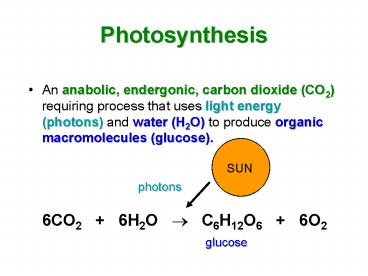
Photosynthesis
- An anabolic, endergonic, carbon dioxide (CO2)
requiring process that uses light energy
(photons) and water (H2O) to produce organic
macromolecules (glucose). - 6CO2 6H2O ? C6H12O6 6O2
2
Breakdown of Photosynthesis
- Two main parts (reactions)
- 1. Light Reaction or
- Light Dependent Reaction
- Produces energy from solar power (photons) in
the form of ATP and NADPH. - 2. Dark Reaction (Calvin Cycle or
- Light Independent Reaction or
- Carbon Fixation or C3 Fixation)
- Uses energy (ATP and NADPH) from light reaction
to make sugar (glucose).
3
- Ch. 13 Photosynthesis
- Annual production of photosynthesis 6 x 1016 g
of C - Occurs in savannahs, forest, ocean, ice
- Evolution 3.5 billion years ago first
photosynthetic bacteria (purple and green sulfur
bacteria) - 2.5 billion years ago
cyanobacteria ?dramatic increase of atmosphereic
oxygen - Contents
- Chloroplasts
- Electron transporting complexes
- Fixation of carbon dioxide
Photosynthesis in context.
Cyanobacteria and purple sulfur bacteria.
4
- Chloroplasts and solar energy
Where Does Photosynthesis Occur?
5
- Chloroplasts and solar energy
- Chloroplasts
- ? the place for photosynthesis to occur
- ? contains its own DNA coding for 100-200
chloroplast proteins (nuclear genes 1000 for
photosynthesis) - ? enclosed by a porous outer membrane and
ion-impermeable inner membrane - ? stroma inner space, rich in enzymes
- ? thylakoid membraneous structure in stroma,
folds into stacks of flattened vesicles
(thylakoid lumen), energy transducing reactions
occur in thylakoid membrane
The chloroplast.
6
Light Dependent Reactions Require Sunlight to
Convert Light Energy into Chemical Energy This
Chemical Energy is Stored in ATP and NADPH
The Thylakoid Membranes contain all the pigments
and all the enzymes necessary for the light phase
of photosynthesis.
7
- How do plants absorb light?
- Light Photon (particle wave)
- 1) Plancks law
- 2) Photoreceptors light absorbing groups in
chloroplasts - ? chlorophyll the principal photoreceptor,
absorbs both blue and red light - ? carotenoid (red) absorbs blue light
- ? Phycocyanin absorbs lower wavelength red
light, common in aquatic system - 3) Light absorption ? electron moves to
high-energy orbital (excited molecule) ? return
to low-energy (ground) state by several
mechanisms - ?
- ?
- ?
- ?
- 4) Exciton transfer and photooxidation
8
Figure 13.05 Visible light absorption by some
photosynthetic pigments.
Figure 13.04 Some common chloroplast
photoreceptors.
9
Figure 13.06 Dissipation of energy in a
photoexcited molecule.
10
- Light harvesting complexes
- Reaction center chlorophyll molecules in complex
where the primary reactions of photosynthesis
occur - 2) Light harvesting complexes extra (antenna)
pigments other than reaction centers - A) Function in photosynthesis
- ? remarkable regular geometry essential for the
light harvesting function - ? different wavelengths
- ? exciton transfer brings the energy to the
reaction center - B) Function to dissipiate excess solar energy as
heat - C) Function as photosensors to regulate plants
growth rate and shape and to coordinate plants
activities according to daily or seasonal light
levels
Figure 13.08 Function of light-harvesting
complexes.
A light-harvesting complex from Rhodopseudomonas
acidophila.
11
- Circadian rhythm
- an approximate daily periodicity, a
roughly-24-hour cycle in the biochemical,
physiological or behavioural processes of living
beings, including plants, animals, fungi and
cyanobacteria - endogenously generated, and can be entrained by
external cues. - allow organisms to anticipate and prepare for
precise and regular environmental changes - - Different Kinds of Biological Rhythms
- 1. Circadian rhythm
- 2. Infradian/ultradian Rhythm
- 3. Circannual Rhythms
- Endogenous factors Suprachiasmatic
- nucleus
(SCN) - the amount of light entering the eye
- pineal gland
- melatonin
12
2.The light reactions 1) Plant, cyanobacteria
two photosynthetic reaction centers
Excitation of reaction centers ? a series of
oxidation-reduction reactions ? oxidation of
water, reduction of NADP ? generation of a
transmembrane proton gradient 2) Photosynthetic
bacteria one photosynthetic reaction
centers Phyosystem II is a light-activated
oxidation-reduction enzymes ? the first light
reaction ? integral membrane proteins (gt 17
subunits in cyanobacteria), numerous prosthetic
groups ? gt 50 chlorophyll molecules for internal
antennas, funneling energy to the two reaction
centers (contains a chlorophyll P680) ? p680
excited by solar energy and give up one electron
(oxidized state) and reduced by electrons derived
from water ? Plastoquinone accept two electron
and soluble in the thylakoid membrane
13
Structure of Photosystem II.
Figure 13.10 Arrangement of prosthetic groups
in Symechococcus Photosystem II.
14
The oxygen-evolving complex of photosystem II
oxidizes water
Oxygen evolving center a lumenal portion of
photosystem II which provides electrons from
water oxidation to reduce P680 ? 2 H2O ? O2
4H 4e- ? 50 O2 production per second per
photosystem II (generates most atmospheric
oxygen) ? catalyst Mn4CaCl1-2Ox(HCO3)y, unusual
inorganic component, but common to all
photosystem II ? 4 H ? released to thylakoid
lumen (drop of pH) ? 4e- tyrosine radical
transfers 4e- to P680 p680 the most powerful
biological oxidant (reduced potential 1.15V)
Reduction potential and electron flow in
Photosystem II.
15
The oxygen-evolving complex of photosystem II
oxidizes water
2 H2O 2 PQ 4H stroma ? O2 4Hlumen
2PQH2 p680 the most powerful biological
oxidant (reduced potential 1.15V)
Figure 13.13 Photosystem II function.
16
Cytochrome b6f links photosystem I and II ? a
second membrane-bound protein complex for
elrctrons to reach resembles mitochondrial
complex III ? eight subunits for monomer
(dimer) 3 subunits (cytochrome b6, cytochrome f
and a Rieske iron-sulfur protein) bearing
electron prosthetic groups ? structure similar to
mitochondria cytochrome b and c, but contains a
chlorophyll and beta carotene (regulator) ?
electron flow cyclic pattern similar to the
mitochondrial Q cycle ? plastocyanin final
electron acceptor, peripheral membrane protein,
transfer electron to Photosystem I
17
Figure 13.15 Plastocyanin.
Figure 13.14 Comparison of transmembrane domains
of cytochrome b6f and mitochondrial cytochrome
bc1.
18
Cytochrome b6f links photosystem I and II ? four
protons released into thylakoid lumen/two
electrons ? pH gradient is the energy for ATP
generation 4 PQH2 4 Plastocyanin(Cu2) 4H
stroma ? 2 PQH2 2 PQ 4 Plastocyanin(Cu)
8Hlumen
Figure 13.16 Cytochrome b6f function.
19
- A second photooxidation occurs at Photosystem I
- ? a large protein complex containing multiple
pigment molecules - ? cyanobacteria a symetric trimer with 31
transmembrane protein helices, 96 chlorophylls
and 22 carotemoids - ? core P700 (relatively high redox potential,
but if exicted, redox potential is very low
(-1.3V)) contains 4 additional chlorophylls,
quinones, iron-sulfur clusters - ? ferredoxin final electron acceptor, one
electron reduction, stromal side protein - ? two electro-transport pathways
- Noncyclic electron flow Z scheme (fig 13-21), no
proton gradient increase, synthesis of 2 NADPH
for 1 O2) - cyclic electron flow (fig 13-22) proton gradient
generation (2 protons for 1 electron)
20
Figure 13.18 Prosthetic groups in Photosystem I.
Figure 13.17 Structure of Photosystem I from
Synechococcus
Figure 13.19 Ferredoxin.
21
- A second photooxidation occurs at Photosystem I
- ? a large protein complex containing multiple
pigment molecules - ? cyanobacteria a symetric trimer with 31
transmembrane protein helices, 96 chlorophylls
and 22 carotemoids - ? core P700 (relatively high redox potential,
but if exicted, redox potential is very low
(-1.3V)) contains 4 additional chlorophylls,
quinones, iron-sulfur clusters - ? ferredoxin final electron acceptor, one
electron reduction, stromal side protein - ? two electro-transport pathways
- Noncyclic electron flow Z scheme (fig 13-21), no
proton gradient increase, synthesis of 2 NADPH
for 1 O2) - cyclic electron flow (fig 13-22) proton gradient
generation (2 protons for 1 electron)
22
Figure 13.21 The Z-scheme of photosynthesis.
Figure 13.20 Noncyclic electron flow through
Photosystem I.
23
Figure 13.22 Cyclic electron flow.
24
Summation of Light Dependent Rxn
(Lumen)
25
Photophosphorylation ATP synthesis by
chemiosmosis ? coupling of dissipiation of pH
gradient to the phosphorylation of ADP ? CF1CF0
complex proton translocating integral membrane
component (CF0) and a soluble CF1 component ? 8
photons absorbed, 12 protons generation? 3 ATP
and one oxygen molecule synthesis
Figure 13.23 Photophosphorylation.
26
Chemiosmosis
27
3. Carbon fixation Dark reaction to fix
atmospheric carbon dioxide in biologically useful
organic molecules (glucose) Rubisco catalyzes
CO2 fixation ? Rubisco ribulose bisphosphate
carboxylase/oxygenase CO2 ribulose 1,
5-bisphosphate (5 C) ? 2 X 3-phosphoglycerate
(3C) ? half of the chloroplasts protein
content ? 3 CO2 fixation per second ? bacteria a
small dimer plant a large multimer (8 large
and 8 small), NOT alosteric, only efficient
packaging? ? acts as also oxygenase
ribulose 1, 5-bisphosphate O2 ?
3-phosphoglycerate (3C) 2-phosphoglycolate
(2C) ? 2-phosphoglycolate substrate for
photorespiration (a pathway to consume ATP and
NADPH and produced CO2, to waste some free energy
of photons when CO2 is not enough) C4 pathway
box 13-B
28
Figure 13.24 The rubisco carboxylation reaction.
29
Figure 13.25 Bacterial and plant rubisco.
30
3. Carbon fixation Rubisco catalyzes CO2
fixation ? Rubisco ribulose bisphosphate
carboxylase/oxygenase CO2 ribulose 1,
5-bisphosphate (5 C) ? 2 X 3-phosphoglycerate
(3C) ? half of the chloroplasts protein
content ? 3 CO2 fixation per second ? bacterial
a small dimer plant a large multimer (8
large and 8 small), NOT alosteric, only efficient
packaging? ? acts as also oxygenase
ribulose 1, 5-bisphosphate O2 ?
3-phosphoglycerate (3C) 2-phosphoglycolate
(2C) ? 2-phosphoglycolate substrate for
photorespiration (a pathway to consume ATP and
NADPH and produced CO2, to waste some free energy
of photons when CO2 is not enough) C4
pathwaybox 13-B
31
Box 13-B
32
The Calvin cycle ? origin of ribulose
1,5-bisphosphate ribulose
5-bisphosphate ATP ? ribulose
1,5-bisphosphate ADP (by phosphoribulokinase)
? 3-phosphoglycerate ATP ? 1,
3-Bisphosphoglycerate ADP
(by phosphoglycerate kinase) ? 1,
3-Bisphosphoglycerate NADPH ?
glyceraldehyde-3-phosphate NADP Pi (by
glyceraldehy-3-phosphate dehydrogenase) ?
glyceraldehyde-3-phosphate siphoned for glucose
or amino acid synthesis ? 3 CO2 9ATP6 NADPH ?
glyceraldehyde-3-phosphate 9 ADP 8 Pi 6
NADP ? quantum yield
33
(No Transcript)
34
- Regulation of carbon fixation
- dark reactions dont occur in the dark stop
during night - regulation of Calvin cycle linked to
availability of light energy - Mg2 ion in the rubisco active site coordinated
in part by carboxylated Lys (activating
CO2)occur at high pH - ?Mg2 activator of Rubisco and other Calvin
cycle enzymes, - rise in stromal pH (to compensate charge
difference due to proton movement) - ?high ratio of reduced ferredoxin
35
Carbohydrate synthesis ? starch i) synthesized
in chloroplast stroma from 3C carbon sugars
generated by the Calvin cycle (and also seeds,
leaves, roots) ii) overall reaction is driven by
the exergonic hydrolysis of the PPi
Glucose-1-phosphate ? ADP-glucose ? starch
36
- Carbohydrate synthesis
- ? sucrose (disaccharide)
- i) synthesized in the cytosol
- ii) glyceraldehyde-3-phosphate transported from
chloroplast to cytosol by an antiport protein - UDP-glucose fructose-6-phosphate ?
sucrose-6-phosphate ? sucrose - iii) sucrose transported to other plant tissues
- ? cellulose
- the other major plant polysaccharide
- synthesized from UDP-glucose
- Cell walls almost-crystalline cables of 36
cellulose polymers
37
Experiment
- ION EXCHANGE CHROMATOGRAPHY
38
The major problem in protein purification
Maximize yield get as many red marbles as
possible High purity take only the red
marbles. These two are opposing forces.
39
(No Transcript)
40
(No Transcript)
41
Exploiting chemical properties of
proteins Purification
- Purification procedures attempt to maintain the
protein in native form. Although some proteins
can be re-natured, most cannot! - To purify a protein from a mixture, biochemists
exploit the ways that individual proteins differ
from one another. They differ in - Size, charge, tag
- Thermal stability
Precipitation with ammonium sulfate (salting out)
solubility
For most protein purifications, all steps are
carried out at 5C to slow down degradative
processes.
Ammonium sulfate precipitation is cheap, easy,
and accommodates large sample sizes. It is
commonly one of the first steps in a purification
scheme.
42
(No Transcript)
43
Size exclusion chromatography
Porous beads made of different but controlled
sizes. Smaller proteins go in and out of beads
and will be retained in the resin. Large
proteins will only go into large beads and will
be retained less. Very large proteins will not
go into any of the beads (exclusion limit). Can
be used as a preparative method or to determine
the molecular weight of a protein in solution.
44
Affinity chromatography
A ligand with high affinity to the proteinis
attached to a matrix. Protein of interest bin ds
to ligand and is retained by resin. Everything
else flows through. Can use excess of the
soluble ligand to elute the protein.
45
Ion exchange chromatography
Anion exchange Column is postively charged
(can bind negativey charged proteins). Cation
exchange Column is negativey charged (can bind
negatively charged proteins).
Exploit the isoelectric point of a protein to
separate it from other macromolecules.
46
(No Transcript)
47
- Ion-Exchange Processes
- Based on exchange equilibria between ions in
solution and - ions of like charge on surface of essentially
insoluble, high- - molecular weight solid.
- Most common cation exchangers
- The strong acid sulfonic acids, SO3-H
- The weak acid carboxylic acids, COOH
- Most common anion exchangers
- The strong base ternary amines, -N(CH3)3OH-
- The weak base primary amines, -NH3OH
48
Ion-exchange chromatography
49
Dissociation of RNAP and purification of s by
ion-exchange chromatography
Carboxymethyl- (-CO2-2) or phospho- (-PO3-2)
cellulose
Fraction number
50
- IEC in practice
- Choose the matrix according to your target
protein - 2. Equilibrate (low salt, lt 20 mM )
- 3. Inject protein sample (in low salt), balance
(wash) - 4. Apply gradient (increasing salt) to elute
proteins - Obey buffer instructions
- a. gradient increasing salt gradient (0-1M NaCl
in 20 mM buffer) - or pH gradient (ampholytes in
chromatofocussing) - b. type of gradient linear gradient /step wise
51
????-???????? ?? ????? ??
??? ????-???????? ????? ???? ?? ??? ??, ? ?????
(ion exchanger)? ?? ?? ?? ?? ?? ???? ????? ????
??
52
????-???????? ?? ????? ??
?Ninhydrin ?? Ninhydrin (triketohydrindene
hydrate)? ????? ???? ???? ?? ??? ???. ?? ???
????? ??? ???? ???. ? ???? NH3? CO2?
???. Ninhydrin (triketohydrindene hydrate) is a
chemical used to detect ammonia or primary and
secondary amines. When reacting with these free
amines, a deep blue or purple color known as
Ruhemann's purple is evolved. Ninhydrin is most
commonly used to detect fingerprints, as amines
left over from peptides and proteins ( or lysine
residues) sloughed off in fingerprints react with
ninhydrin
53
????-???????? ?? ????? ??
- ??? ?? ? ??
- 0.1 N HCl, 0.2 M pH 8.2 Tris-HCl (????), ????
??(0.2 acetone ??), ???????? ?, ?? ?? ??, ???,
??, ?? ???, ?????, ??, ???? ?? ( Aspartate,
Methionine, Arginine ) - ?????
- 1. 0.1 N HCl 10ml? ??? ????? ??? ?? 12cm ????
???. - ( ??? ?? ??? ?? ??? ?? ??? ?? ??? ?? ??? ??.)
- 2. 0.2 ml? ???? ????? ?? ? ??? ??? ??? ???? ??.
- 3. 0.2 ml? 0.1 N HCl? ??? ???? ?? ??? ??, ? ??? ?
? ????. - 4. 2 ml? 0.1 N HCl? ?? ? ?? ??? 0.1 N HCl 500 ml?
?? ???? ????, ???? ?? ???? ??? ???. - 5. ????? ???? ??? ??? 5?? ???? ??? ????? ??? ??
????? ??? ??? ?? ???? ??? ????? ????. - 6. 0.2 M Tris-HCl ???? 2ml? ?? ???? ? ?? ????? ?
?? ????? ??? ??? ??? ????? ?? ????. - 7. ???? ??? ?? ????? ????.
54
Problem 13.20
55
Page 398
56
Page 416b
57
Page 405