Introduction to the Chemistry of NitrogenContaining Compounds: Amines - PowerPoint PPT Presentation
1 / 63
Title:
Introduction to the Chemistry of NitrogenContaining Compounds: Amines
Description:
A nitrogen-based link is vital in the polymeric molecules known as proteins and peptides. ... Atropine, scopolamine and cocaine: tropane alkaloids. Fig. 20.84 ... – PowerPoint PPT presentation
Number of Views:1282
Avg rating:3.0/5.0
Title: Introduction to the Chemistry of NitrogenContaining Compounds: Amines
1
Introduction to the Chemistry of
Nitrogen-Containing Compounds Amines
- Introduction (page 1011)
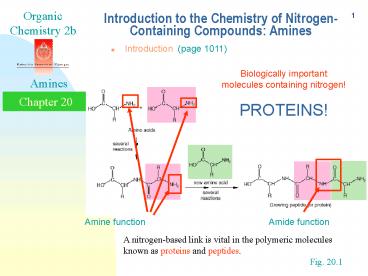
Biologically important molecules containing
nitrogen! PROTEINS!
2
Introduction to the Chemistry of
Nitrogen-Containing Compounds Amines
- Introduction (page 1012)
Biologically important molecules containing
nitrogen! ALKALOIDS
3
Introduction to the Chemistry of
Nitrogen-Containing Compounds Amines
- Introduction (page 1012)
IMPORTANT HARD DRUGS ARE AMINES!!!
Amines!
Some alkaloids-nitrogen containing molecules with
great biological activity. Fig. 20.2
4
Introduction to the Chemistry of
Nitrogen-Containing Compounds Amines
- 20.1 Nomenclature (page 1012)
Substituted amines. Fig. 20.3
5
Introduction to the Chemistry of
Nitrogen-Containing Compounds Amines
- 20.1 Nomenclature (page 1012)
Examples
A naming scheme for primary, secondary and
tertiary amines. Fig. 20.4
6
Introduction to the Chemistry of
Nitrogen-Containing Compounds Amines
- 20.1 Nomenclature (page 1013)
Name these amines!
A naming scheme for primary, secondary and
tertiary amines. Fig. 20.4
7
Introduction to the Chemistry of
Nitrogen-Containing Compounds Amines
- 20.1 Nomenclature (page 1013)
A naming scheme for primary, secondary and
tertiary amines. Fig. 20.4
8
Introduction to the Chemistry of
Nitrogen-Containing Compounds Amines
- 20.1 Nomenclature (page 1013)
Naming amines as substituted alkanes necessary
for more complex molecules!
Still another method for naming amines. Fig. 21.6
9
Introduction to the Chemistry of
Nitrogen-Containing Compounds Amines
- 20.1 Nomenclature (page 1013)
Name these amines!
10
Introduction to the Chemistry of
Nitrogen-Containing Compounds Amines
- 20.1 Nomenclature (page 1013)
Name these amines!
Still another method for naming amines. Fig. 21.6
11
Introduction to the Chemistry of
Nitrogen-Containing Compounds Amines
- 20.2 Structure and Physical Properties of
Amines (page 1016)
Why shorter than C-C and longer than C-O?
The carbon-nitrogen bond in amines is shorter
than a normal carbon-carbon bond. Fig. 20.14
12
Introduction to the Chemistry of
Nitrogen-Containing Compounds Amines
- 20.2 Structure and Physical Properties of
Amines (page 1016)
If true what could be the bond distance of CH3-F?
See 16.2 for reasoning!
The carbon-nitrogen bond in amines is shorter
than a normal carbon-carbon bond. Fig. 20.14
13
Introduction to the Chemistry of
Nitrogen-Containing Compounds Amines
- 20.2 Structure and Physical Properties of
Amines (page 1016)
Problem 20.1 Why is the carbon-nitrogen bond
distance in aniline slightly shorter than that in
methylamine?
14
Introduction to the Chemistry of
Nitrogen-Containing Compounds Amines
- 20.2 Structure and Physical Properties of
Amines (page 1017)
Sp3 hybridization at nitrogen and thus pyramidal!
Some bond angles in simple amines Fig. 20.16
15
Introduction to the Chemistry of
Nitrogen-Containing Compounds Amines
- 20.2 Structure and Physical Properties of
Amines (page 1017)
Why not exactly 109.5o?
Some bond angles in simple amines Fig. 20.16
16
Introduction to the Chemistry of
Nitrogen-Containing Compounds Amines
- 20.2 Structure and Physical Properties of
Amines (page 1017)
What happens here? Why not for carbon?
Amine inversion interconverts enantiomeric
amines Fig. 20.17
17
Introduction to the Chemistry of
Nitrogen-Containing Compounds Amines
- 20.2 Structure and Physical Properties of
Amines (page 1017)
Amine inversion has a planar transition
state. Fig. 20.18
18
Introduction to the Chemistry of
Nitrogen-Containing Compounds Amines
- 20.2 Structure and Physical Properties of
Amines (page 1017)
Amine inversion has a planar transition
state. Fig. 20.18
19
Introduction to the Chemistry of
Nitrogen-Containing Compounds Amines
- 20.2 Structure and Physical Properties of
Amines (page 1018)
What if the amine is chiral?
20
Introduction to the Chemistry of
Nitrogen-Containing Compounds Amines
- 20.2 Structure and Physical Properties of
Amines (page 1018)
Problem 20.4 In contrast to simple amines,
aziridines (three-membered rings containing a
nitrogen) can often be separated into
enantiomers. For example, the activation energy
for the inversion of 1,2,2-trimethylaziridine is
about 18.5 kcal/mole, much higher than for simple
amines. Explain.
Fig. 20.20
21
Introduction to the Chemistry of
Nitrogen-Containing Compounds Amines
- 20.2 Structure and Physical Properties of
Amines (page 1018)
replace by phenyl
Problem 20.5 If there is a phenyl group attached
to the nitrogen atom of the aziridine, the
barrier to inversion decreases.
22
Introduction to the Chemistry of
Nitrogen-Containing Compounds Amines
- 20.2 Structure and Physical Properties of
Amines (page 1018)
replace by phenyl
Problem 20.5 If there is a phenyl group attached
to the nitrogen atom of the aziridine, the
barrier to inversion decreases. Explain.
23
Introduction to the Chemistry of
Nitrogen-Containing Compounds Amines
- 20.2 Structure and Physical Properties of
Amines (page 1020) - Decomposing fish owes its characteristic
unpleasant nature to amines which are liberated
in this process.
Problem 20.6 Many people use lemon when eating
fish. This custom is a carryover from the days
when it was difficult to preserve fish, and the
lemon acted to diminish the unpleasant odor (if
not the decomposition). Lemons contain 5-8
citric acid and this contributes to their soure
taste. Explain why lemon juice is effective at
reducing the odor of fish.
24
Introduction to the Chemistry of
Nitrogen-Containing Compounds Amines
- 20.4 Acid and Base Properties of Amines (page
1021)
Ammonia acting as a Brønsted base and as a
nucleophile Fig. 20.24
25
Introduction to the Chemistry of
Nitrogen-Containing Compounds Amines
- 20.4 Acid and Base Properties of Amines (page
1022)
Ammonium ions with high pKa values are related to
strongly basic amines, and ammonium ions with low
pKa values are related to weakly basic
amines. Fig. 20.25
26
Introduction to the Chemistry of
Nitrogen-Containing Compounds Amines
- 20.4 Acid and Base Properties of Amines (page
1022)
The pKa values for some simple ammonium
ions Table 20.2
27
Introduction to the Chemistry of
Nitrogen-Containing Compounds Amines
- 20.4 Acid and Base Properties of Amines (page
1022)
The gas-phase acidity of ammonium ions. Figure
20.26
28
Introduction to the Chemistry of
Nitrogen-Containing Compounds Amines
- 20.4 Acid and Base Properties of Amines (page
1022)
The gas-phase acidity of ammonium ions. Figure
20.26
29
Introduction to the Chemistry of
Nitrogen-Containing Compounds Amines
- 20.4 Acid and Base Properties of Amines (page
1024)
In solution, amines are stabilized through
dipole-dipole interactions and hydrogen bonding.
Large groups attached to nitrogen will interfere
with these stabilized interactions. Figure 20.27
30
Introduction to the Chemistry of
Nitrogen-Containing Compounds Amines
- 20.4 Acid and Base Properties of Amines (page
1024)
Problem 20.8
Amines can be stabilized by other factors.
Explain the pKa data in Figure 20.28
Figure 20.28
31
Introduction to the Chemistry of
Nitrogen-Containing Compounds Amines
- 20.4 Acid and Base Properties of Amines (page
1024)
Why?
Amines are much stronger bases than alcohols and
the corresponding ammonium ions are much weaker
acids than oxonium ions. Fig. 20.30
32
Introduction to the Chemistry of
Nitrogen-Containing Compounds Amines
- 20.4 Acid and Base Properties of Amines (page
1025)
Problem 20.9 The stability of oxonium ions
depends on the nature of the negatively charged
counterion. Fluoroborate (BF4-) is an especially
favorable counterion. Why?
33
Introduction to the Chemistry of
Nitrogen-Containing Compounds Amines
- 20.4 Acid and Base Properties of Amines (page
1025)
?
?
Primary and secondary amines are weak Brønsted
acids. Fig. 20.31
34
Introduction to the Chemistry of
Nitrogen-Containing Compounds Amines
- 20.4 Acid and Base Properties of Amines (page
1025)
Primary and secondary amines are weak Brønsted
acids. Fig. 20.31
35
Introduction to the Chemistry of
Nitrogen-Containing Compounds Amines
- 20.4 Acid and Base Properties of Amines (page
1025)
Very strong bases such as alkyllithium reagents
can remove a proton from an amine to give an
amide ion. Bu
CH2CH2CH2CH3 Fig. 20.32
36
Introduction to the Chemistry of
Nitrogen-Containing Compounds Amines
- 20.4 Acid and Base Properties of Amines (page
1025)
The amide ion is a far stronger base (and
nucleophile) than the parent amine. Fig. 20.33
37
Introduction to the Chemistry of
Nitrogen-Containing Compounds Amines
- 20.5 Reactions of Amines (page 1026)
- 20.5a Alkylation of Amines
38
Introduction to the Chemistry of
Nitrogen-Containing Compounds Amines
- 20.5 Reactions of Amines (page 1026)
- 20.5a Alkylation of Amines
39
Introduction to the Chemistry of
Nitrogen-Containing Compounds Amines
- 20.5 Reactions of Amines (page 1026)
- 20.5a Alkylation of Amines
40
Introduction to the Chemistry of
Nitrogen-Containing Compounds Amines
- 20.5 Reactions of Amines (page 1026)
- 20.5a Alkylation of Amines
The formation of an ammonium ion through
alkylation of a tertiary amine Fig. 20.35
41
Introduction to the Chemistry of
Nitrogen-Containing Compounds Amines
- 20.5 Reactions of Amines (page 1027)
- 20.5b Additions of Amines to Carbonyl Groups
42
Introduction to the Chemistry of
Nitrogen-Containing Compounds Amines
- 20.5 Reactions of Amines (page 1027)
- 20.5b Additions of Amines to Carbonyl Groups
Imine and enamine formation from reactions of
amines with carbonyl compounds. Fig. 20.36
43
Introduction to the Chemistry of
Nitrogen-Containing Compounds Amines
- 20.7 Alkaloids (page 1047)
Basic (alkaline) compounds extracted from natural
sources (plants) generally amines!
Usually chiral compounds and single enantiomers!
Some have powerful biological activity!
44
Introduction to the Chemistry of
Nitrogen-Containing Compounds Amines
- 20.7 Alkaloids (page 1047)
Basic (alkaline) compounds extracted from natural
sources (plants) generally amines!
A scheme for isolation of alkaloids from plant
material. Fig. 20.78
45
Introduction to the Chemistry of
Nitrogen-Containing Compounds Amines
- 21.7 Alkaloids (page 1109)
Usually chiral compounds and single enantiomers!
46
Introduction to the Chemistry of
Nitrogen-Containing Compounds Amines
- 20.7 Alkaloids (page 1048)
Some have powerful biological activity!
47
Introduction to the Chemistry of
Nitrogen-Containing Compounds Amines
- 20.7 Alkaloids (page 1048)
Some alkaloids containing various nitrogen
heterocycles. Fig. 20.80
48
Introduction to the Chemistry of
Nitrogen-Containing Compounds Amines
- 20.7 Alkaloids (page 1048)
Morphine
The structure of morphine. Fig. 20.81
49
Introduction to the Chemistry of
Nitrogen-Containing Compounds Amines
- 20.7 Alkaloids (page 1048)
Find the differences!
50
Introduction to the Chemistry of
Nitrogen-Containing Compounds Amines
- 20.7 Alkaloids (page 1048)
Reaction necessary for structure elucidation of
morphine, codeine etc.
A Hofmann elimination applied to
codeine. Fig. 20.82
51
Introduction to the Chemistry of
Nitrogen-Containing Compounds Amines
- 20.7 Alkaloids (page 1048)
Problem 20.25 Write arrow formalisms for the
reactions of Figure 20.82
A Hofmann elimination applied to
codeine. Fig. 20.82
52
Introduction to the Chemistry of
Nitrogen-Containing Compounds Amines
- 20.5 Reactions of Amines (page 1028)
- 20.5c Elimination Reactions of Amines
Hofmann elimination reactions of amines compared
to dehydration reactions. Fig. 20.39
53
Introduction to the Chemistry of
Nitrogen-Containing Compounds Amines
- 20.5 Reactions of Amines (page 1028)
- 20.5c Elimination Reactions of Amines
Problem 20.12 Provide mechanisms for the
reactions of Figure 20.40
Fig. 20.40
54
Introduction to the Chemistry of
Nitrogen-Containing Compounds Amines
- 20.7 Alkaloids (page 1049)
A non-natural derivative of morphine
What is the name of this well-known hard-drug?
Heroin!!!
Fig. 20.83
55
Introduction to the Chemistry of
Nitrogen-Containing Compounds Amines
- 20.7 Alkaloids (page 1049)
Non-natural derivative of morphine
Morphine
Hardly addictive
Addictive!
Codeine and heroin are derivatives of
morphine. Fig. 20.83
56
Introduction to the Chemistry of
Nitrogen-Containing Compounds Amines
- 20.7 Alkaloids (page 1049)
Non-natural derivative of morphine
Morphine
Problem 20.26 Unfortunately, morphine is easily
converted into heroin, even in a basement lab.
Suggest a way to achieve this conversion.
57
Introduction to the Chemistry of
Nitrogen-Containing Compounds Amines
- 20.7 Alkaloids (page 1049)
From coca leaves an anesthetic.
Medicine pupil dilation inhibition of smooth
muscle spasms
Causes twilight sleep, a state of analgesia and
amnesia.
58
Introduction to the Chemistry of
Nitrogen-Containing Compounds Amines
- 20.7 Alkaloids (page 1050)
Indole Alkaloids
neurotransmitter
59
Introduction to the Chemistry of
Nitrogen-Containing Compounds Amines
- 20.7 Alkaloids (page 1050)
Indole Alkaloids
Powerful effects on brain and perception
(hallucination)
neurotransmitter
60
Introduction to the Chemistry of
Nitrogen-Containing Compounds Amines
- 20.7 Alkaloids (page 1050)
Problem 20.28 The reactions in Figure 20.87 were
important in the synthesis of LSD. Provide
structures for the indicated compounds.
Fig. 20.87
61
Introduction to the Chemistry of
Nitrogen-Containing Compounds Amines
- 20.8 Alkaloids (page 1051)
A powerful antimalarial agent
Quinine Fig. 20.88
62
Introduction to the Chemistry of
Nitrogen-Containing Compounds Amines
- 20.8 Alkaloids (page 1051)
Problem 20.29 Outline mechanisms for the
following transfromations leading to quinine
(Fig. 20.89).
Fig. 20.89
63
Introduction to the Chemistry of
Nitrogen-Containing Compounds Amines
- 20.12 Additional Problems (page 1058)
- Problems 20.30 20.31 20.32 20.33 21.36 21.39
c,d,e,g,h.